Survivorship and rehabilitation:
Policy recommendations for quality improvement in cancer survivorship and rehabilitation in EU Member States
This is a chapter Survivorship and rehabilitation (Cancon work package 8) of the Guide. See the Annexes.
See the full Guide and other chapters as pdf's.
Cancon Guide is the main delivery of the joint action.
Contents of Survivorship and rehabilitation:
Introduction
Methods
Results
Perspectives in survivorship and rehabilitation cancer research
Conclusions and recommendations
References
Main messages
- Cancer survivors’ follow-up, late effect management and tertiary prevention needs to be anticipated, personalized and implemented into care pathways, with active participation of survivors and relatives.
- Improvement of early detection of patients’ needs and their access to rehabilitation, psychosocial and palliative care services is required.
- An integrated and multiprofessional care approach with a coordination of community care providers and services are needed to implement a survivorship care plan that enhances patient’s self-management and quality of life.
- For children, adolescents and young adults survivors, late health and psychosocial effects of cancer and its treatments need to be anticipated and addressed.
- More research in the area of survivorship is needed to provide data on late effects, as well as the impact and cost-effectiveness of supportive care, rehabilitation, palliative and psychosocial care interventions.
Introduction
The new cancer survivorship challenge: going beyond quality of care and ensuring quality of life
Over past decades, the number of cancer survivors has increased substantially in Europe as well as in most high-income countries, as a result of the ageing population, progress in early diagnosis and effectiveness of therapies. Survival from cancer is improving and the five-year global prevalence of all cancers in Europe was about 9.7 million people in 2012 (1).
However cancer survival still varies widely within Europe, with lower rates in eastern countries compared with Nordic and central European countries (2).
Whether being cured (disease-free) or not, cancer survivors do experience late and long-term effects of treatment, emotional distress and potentially tumour recurrence. These effects represent challenges for health care systems, which have to ensure their appropriate follow-up care and quality of life: moving from “how long” people live after diagnosis to “how well” people can expect to live from diagnosis onward.
Recognizing this, the Council of the European Union invited Member States to “take into account the psycho-social needs of patients and improve the quality of life for cancer patients through support, rehabilitation and palliative care” (3). Eurostat defines quality of life as “the functional capabilities that citizens should have available to effectively pursue their self-defined well-being, according to their own values and priorities” (4). The patient-centred approach is, therefore, of high importance in the planning of (any type of) care provision.
Many studies have investigated the issues in the follow-up care of cancer survivors (5–7) and found the most impeding factors for the quality of cancer follow-up care were poor coordination of care, lack of communication among health care providers, uncertainties about “who is responsible” for the follow-up care and occurrence of many psychosocial unmet needs.
Based on the Institute of Medicine recommendations (5), the (site-specific) templates developed by the American Society of Clinical Oncology for the follow-up care of cancer survivors recommended the use of survivorship care plans to overcome these issues (8). Despites great values, survivorship care plans are still scarcely used because of two main barriers (7,9): the feasibility of integrating them into practice and the human and financial resources required to develop and manage these plans.
At the moment, no clear consensus exists regarding the content, format, management and implementation of long-term follow-up care plans for cancer survivors. This chapter aims at reviewing the existing knowledge and evidence about these plans and at providing policy recommendations for health administrators and policy-makers in charge of cancer control in EU Member States, in order to facilitate or engage in the improvement of the quality of cancer survivor’s care and life.
The definition of cancer survivorship
The cancer care pathway or continuum has evolved alongside treatment innovations, as did attempts to define cancer survivorship. First definitions were based on the three key phases of cancer treatment: patients recently diagnosed, patients who completed their treatment and experience periodic examinations and patients considered as “cured” (10). This can be summarized as “the experience of living with, through and beyond a diagnosis of cancer” (11).
Cancer treatment improvements implied the appearance of patients living cancer free for many years but who experience recurrence or develop second primary cancer, patients with intermittent periods of active disease (chronic cancers), patients living for many years with advanced cancers and those who live after the expected death (12).
For the purpose of this chapter, a cancer survivor is defined as anyone with a diagnosis of cancer and who is still alive. This includes patients having completed primary therapy and who are free of disease as well as those patients living with recurrent and/or advanced disease.
According to WHO (13), rehabilitation (though not cancer specific) is defined as “a process aimed at enabling them [people with disabilities] to reach and maintain their optimal physical, sensory, intellectual, psychological and social functional levels.” In fact, cancer may be seen as a chronic illness with patients enduring physical and psychological symptoms years after treatment.
A new paradigm to be integrated in the cancer care pathway
Several reviews and surveys have been conducted in order to identify the best model of care for improving the cancer survivor’s quality of care and quality of life (5,6,9). The best approach appears to be the use of survivorship care plans that include the provision of medical and nonmedical care.
Two main models are used: the shared care model (see below) and specialized survivorship clinics (7). The choice of which to use mainly depends on the national health care system, including the role of primary care and the reimbursement scheme (insurance coverage).
Even though evidence shows the important added-value for patients, health care providers and health care systems from survivorship care plans, these plans are scarcely used and few cancer patients have access to one.
The provision of policy recommendations for the content, format, management and implementation of survivorship care plans in EU Member States is the main purpose of this chapter.
Methods
Results of the preliminary work on existing guidelines or plans for long-term follow-up care for cancer patients reported that four countries can be recognized as pioneers: the United States, Canada and Australia, which follow the work achieved by the Institute of Medicine (5), and the United Kingdom (14).
Recommendations from the Institute of Medicine (5) for the United States and from the National Cancer Survivorship Initiative for the United Kingdom (14) and the content of their follow-up care plans were compared and five key areas were identified for investigation in the field of long-term follow-up care for cancer patients:
- medical follow-up: management of the late effects and tertiary prevention
- psychological support
- social rehabilitation including employment issues
- empowerment of cancer survivors
- multidisciplinary approach and coordination of care providers.
Four cross-cutting issues received particular attention:
- care for childhood cancer survivors
- inequalities in survivorship
- cancer information and data registration
- research.
In order to collect evidence about these nine issues and to translate it into policy recommendations for EU Member States, a three-fold methodology has been used (see supplemental information on methodology provided at www.cancercontrol.eu).
First, a literature review has been conducted using key words for the search and spread sheets to report outcomes.
Second, a critical appraisal exercise has been organized with a deliberative process involving invited EU experts to discuss the results and their applicability in EU Member States.
Third, in order to have insights into what is in use in EU Member States for cancer follow-up care, a survey was launched and 21 replies from nine EU countries have been analysed and compared (Annex 7.1).
Results
Content of long-term cancer follow-up care plans
A total of 151 publications (see supplemental information provided at www.cancercontrol.eu) have been retained and synthesized in the following to present key evidence related to the management of the late effects, tertiary prevention, psychological support and social rehabilitation in cancer follow-up care.
According to the National Cancer Institute in the United States (15), a survivorship care plan is “a detailed plan for a patient’s follow-up care after treatment for a disease ends. In cancer, the plan is based on the type of cancer and the treatment the patient received. A survivorship care plan may include schedules for physical exams and medical tests … Follow-up care also checks for health problems that may occur months or years after treatment ends … and may also include information to help meet the emotional, social, legal, and financial needs of the patient. It may include referrals to specialists and recommendations for a healthy lifestyle, such as changes in diet and exercise and quitting smoking.”
Medical follow-up: management of late effects
Cancer and its treatment have both direct and indirect effects, such as treatment-induced cardiotoxicity (16–18), bone loss, fatigue, pain, depression, endocrine and fertility problems (19), and these are important elements for follow-up surveillance (7,20). The challenge is to identify the patients at risk of encountering late effects and thereafter use preventive measures to mitigate these effects as much as possible. Deterioration of physical, mental and social quality of life in survivorship is strongly connected to precarious situation (i.e. low income, unemployment and other socially disadvantageous positions) (21).
The evidence in the literature is incomplete for childhood, adolescent and young adult cancer survivorship. For this population, the late and long-term effects that negatively affect their health and well-being include cardiovascular diseases, neurocognitive functioning (22), sexual and reproductive functions (23) and renal and endocrine functions.
Findings suggest that there is a need for deeper understanding of factors associated with increased morbidity susceptibility. This means that elements such as genomics, personalized and behavioural medicine, treatment-related toxicities, psychology, cardiology and endocrinology, as well as genetic predisposition, should be examined (24). The relationships between cancer treatments and the natural ageing process, as well as the interaction of multiple morbidities, are issues that should be further explored (25).
Medical follow-up: tertiary prevention
According to the IARC, tertiary prevention is “the use of treatment and rehabilitation programmes to improve the outcome of illness among affected individuals” (26).
Healthy lifestyle has positive effects on the prevention and management of late effects and cancer recurrence (5,27). Healthy food, adequate physical activity (28,29), avoidance of excessive sun
exposure, limited alcohol consumption, stress reduction (30) and smoking cessation are important elements to consider for increased and quality survival.
Some physical activity is better than none and exercise can safely be undertaken by all cancer patients even during the advanced stage of the disease (31). Physical activity during treatment has shown to increase the percentage of patients completing therapy (32,33). It is effective in reducing both physiological and psychological treatment-related effects; speeds up recovery after treatment; improves pain in neck, shoulder and axillary region in breast cancer (34); and may prevent cancer recurrence (35,36). Incontinence, fatigue, body constitution and quality of life can be improved by physical exercise for patients during and after prostate cancer (37).
Stress is considered a risk factor for cancer recurrence. Stress-management interventions have been proved to reduce mortality and, therefore, may be beneficial in the prevention of recurrence (38,39).
Survivorship care for patients with advanced cancer
Medical advances have enabled cancer patients to live longer with active advanced-stage diseases. Although the symptoms and medical needs are similar to those of disease-free survivors, psychosocial concerns are different (40).
Most publications address the early integration of supportive and palliative care into cancer care pathways of patients diagnosed with advanced cancers (41–44).
Alongside the symptoms and side-effects of cancer treatment (e.g. incontinence, neuropathy, hair loss, nausea), patients with advanced-stage disease have to face physical and psychological well-being fluctuations that have a disruptive impact on their ability and willingness to cope with treatment and to plan for the future (45).
As for disease-free survivors, patients with advanced cancer report many unmet psychosocial needs but also additional unmet information needs, particularly related to euthanasia, living wills, financial and legal issue and hospices (46,47).
Patient-centred approach for long-term follow-up
Multidimensional needs’ assessment as the starting point
The literature review showed that early and systematic detection of needs (Annex 7.2) in psychological support, social and physical rehabilitation, supportive and palliative care are necessary in order to orientate patients towards tailored health care interventions (48–50). It also demonstrated the necessity to anticipate certain issues that the patients and their relatives will face during the survivorship and rehabilitation period.
Measuring health-related quality of life should be integrated as an early, systematic and recurrent step in the long-term follow-up care of cancer survivors (51) (Fig. 7.1). Several tools already exist and can be used routinely.
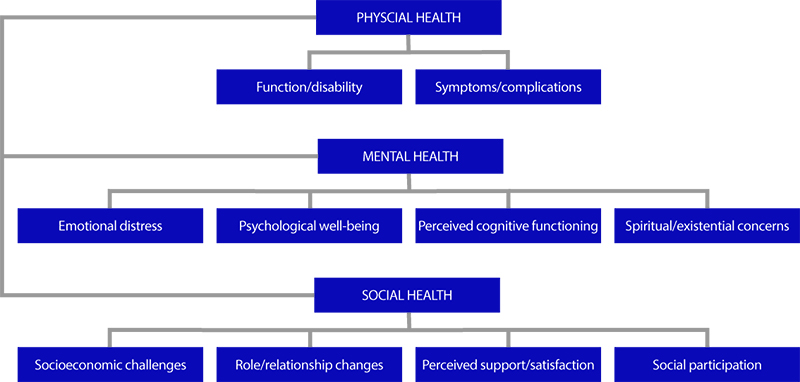
Fig. 7.1 Health-related quality of life in cancer survivors
Source: Victorson et al., 2006 (51).
Psychological support
Diagnosis of cancer often generates major distress for patients and their families. Depression and other symptoms that impact quality of life during the entire cancer pathway are well documented, particularly among breast cancer survivors (52). Reviews highlight problems with fear of recurrence, fatigue, sexual health, depressive symptoms, pain and late or long-term effects because of cancer treatments. Individuals follow various trajectories of psychological adjustment during survivorship (53).
Evidence shows that psychological interventions reduce psychological morbidity and improve patients’ adjustment to illness, quality of care and well-being (54–56). The access to psychological intervention during survivorship can be difficult, either because of patients’ reluctance or because insufficient care is offered (57). Instruments for diagnosing psychosocial conditions, level of distress and psychosocial needs are not yet routinely used in all cancer settings (50,58). Several countries in Europe, the United States, Canada and Australia have developed clinical practice guidelines that assist clinicians in using evidence-based psychosocial care in their practice (49,59–62).
Inequalities are rarely mentioned in the selected articles from the review. However, geographical and social isolation may create greater difficulties in accessing quality psychosocial and palliative care for those living in rural areas and diagnosed with cancer (63). The mapping of psychosocial care resources in Europe shows that the provision of psycho-oncology services is very diverse, irregular and greatly depends on whether or not a country and its national cancer control plan or strategy considers psychosocial care as an element of multidisciplinary cancer care (64). Resources in this area are still scarce and widely variable across countries and type of hospital (cancer centre versus local hospital), mainly because of lack of financing and policy; in some countries they are even non-existent (65).
Professional reintegration
Results include issues such as employment and insurance that cancer patients have to face (5,66,67). The case study based on the VICAN 2 survey (68) illustrates these issues (Box 7.1).
---
Box 7.1 Case study examining employment issues: the VICAN 2 study
Background
The French national study VICAN 2 sheds light on people’s daily life and the impact of cancer during the survivorship and rehabilitation period. This case study focuses on the inequalities on both the occupation rate and income two years after the first diagnosis of cancer.
Methodology
Computer-assisted telephone interviews of 40 minutes duration were conducted between January 2012 and June 2013 with about 4350 patients who had been diagnosed with cancer in 2010.
Outcome: cancer impact on income
At the diagnosis stage in 2010, 20.9% could be considered as poor compared with 14% in the general population (data from the French National Institute for Statistics and Economic Studies). Two years after the diagnosis, the gap had increased, with 25.1% of people living with a cancer being below the poverty threshold compared with 14.3% of the general population.
Losing one’s job is not the only way the diagnosis of cancer has a financial impact on household income: most of the people who continued their employment during cancer diagnosis, treatment and survivorship had to face various important changes in their working time depending on their health status, which had impact on their income.
Outcome: professional situation two years after diagnosis of cancer
The professional situation of people diagnosed with cancer was shown to have considerably deteriorated two years after the diagnosis, with an activity rate decreasing from 88.2% in 2010 to 79.9% in 2012. The employment rate decreased to 61.3% and the unemployment rate was 11.1% (i.e. four points higher compared with 2010).
The most vulnerable people were mainly manual workers; the youngest and oldest; married people; people with an educational level below advanced level (A-level); those with fixedterm precarious working contracts; and those working in small and medium enterprises. This demonstrates a "double penalty", which brings together the unfavourable characteristics of the job market and the impact of the cancer diagnosis.
Most of the people aged between 18 and 57 years had lost their job 15 months after the diagnosis (91.6%) and 21.8% lost it at the moment of diagnosis. The waiting period before finding a new job was 11 months on average (i.e. an additional delay of 6 months compared with data for the general population).
Further research
A similar national study with the same methodology will be launched to assess people’s quality of life 5 years after the diagnosis.
Recommendation
Similar studies should be conducted in different EU countries to better assess the impact of cancer on people’s daily lives in order to address their needs and target sources of inequalities. Source: Institut National du Cancer, 2014 (68).
---
Return-to-work support should be integrated early into the cancer care pathway, exploring the feasibility of adequate or progressive return to work and discussing with employers about working conditions (69,70). Both health care providers and employers have a role to play (63,71,72).
A strong emphasis is put on the positive effects of early psychosocial interventions in supporting cancer survivors with employment-related issues, ideally immediately after diagnosis and during treatment (73). Psychosocial and vocational rehabilitation need to take a person-centred approach based on each individual’s situation: diagnosis and prognosis, medical and nonmedical treatments, intra- and interpersonal factors, patient values, aspirations and priorities, the attitude of colleagues, job demands, and so on. These determinants should be taken into account when planning reintegration into the working environment (70,74–79).
Supportive return-to-work interventions can be directed to employees or to the work environment and employers (80–82). The first approach aims at maintaining or enhancing the employability of cancer survivors. Work environment-directed interventions aim at adapting workplace environment, equipment, tasks and working time patterns to the needs of the cancer survivor.
More evidence is needed on the effectiveness of return-to-work interventions and on work conditions for cancer survivors who do return to work (83).
Regarding employment and return-to-work issues, there are some good examples of regulation for the protection of cancer survivors and their relatives.
An Italian regulation passed in 2003 (decree-law n° 276/2003, article 46, as amendment of decreelaw n° 61/2000, article 12 bis) prescribed the right for cancer patients working in the private sector to switch from full-time to part-time positions while under treatment, and to reverse to full-time according to their needs and capability. The same right was extended to public employees in 2007 (law n° 247/2007, article 1, subsection 44). Within the same legal framework, relatives (caregivers) of cancer patients are given priority over part-time applications as long as there are positions available.
In the United Kingdom, the employment provisions of the Equality Act 2010 protect anyone who has, or has had, a disability (including people affected by cancer). The Act requires employers to make reasonable adjustments for employees with a disability. But it also includes important provisions to prevent discrimination arising from disability, indirect discrimination and discrimination against carers.
Other socioeconomic issues relate to health, disability and life insurances. European surveys (84) and the Institute of Medicine study (5) have reported that psychosocial workers should provide information on the potential insurance, employment and financial consequences of cancer through provision of a directory of cancer-related resources (e.g. online or telephone listings) and/or information in the form of general information brochures. Raising these issues with patients will at least let them know that help is available.
Children, adolescents and young adult survivors of cancer
For children, adolescents and young adults, the psychosocial experience of the illness is highly variable (85,86). It is sometimes years after the cancer is cured that the psychosocial impact of the illness occurs, leading to requests for support and psychosocial care (87,88). The utility of supportive and rehabilitation care has been proved, in particular adapted physical activity for children, to be associated with better health-related quality of life (89,90).
The literature suggests a routine yearly psychosocial assessment with attention to behavioural issues and educational and/or vocational progress to detect early signs of psychosocial suffering (91,92). Parents or relatives need to be involved in every step and are always considered as facilitators if they are properly educated, informed and coached by the health care providers (93).
Management of long-term folllow-up care plans
Results from 55 publications have been retained, synthesized and discussed in order to present the key evidence regarding the management of survivorship care plans. There were three main issues:
the role of multidisciplinary teams and the coordination of providers; the empowerment of cancer survivors; and the self-management perspective.
Multidisciplinary approach and coordination of cancer care providers
The clinical follow-up system as currently applied in survivorship shows low added-value. Multidisciplinary teams often disregard survivorship and rehabilitation issues. The main barriers with regards to their role in undertaking a survivorship care plan is lack of vision regarding redesigning the cancer patients’ pathway and the team’s workload (94). In addition, multidisciplinary teams also feel that they lack time and information about a follow-up plan. There is little evidence in the literature regarding follow-up care and the role of GPs (see Chapter 6).
The literature shows that survivorship care plans can be built upon the clinical management of a multidisciplinary team and could include addressing patients’ late effects of treatment and psychosocial needs with a rehabilitation slant (95–98). Survivorship care plans can be managed according to different models of care coordination with a common starting point of a specialist in a multidisciplinary teams providing follow-up to create a holistic and integrated approach to survivors’ health (70,99,100). Three conditions arise as core practices in the literature:
- GPs or a primary care team should play a relevant role in patients’ follow-up
- the follow-up model should provide a rapid re-entry to specialized cancer care, if required; and
- a health care professional should assume the role of a coordinating case manager by being a point of reference and contact for the patient and the team.
Other components to be taken into account include the possibility of modifying health insurance coverage to include the follow-up as such and facilitating patient’s access to community resources, patients’ support groups and volunteers (101).
As 80% of young people with cancer are now surviving, all relevant stakeholders should be informed on possible risks or late effects of the cancer treatment received. The lack of information on many patients’ medical history becomes particularly critical as children become adults. The Survivorship Passport initiative (102) can provide a solution to this problematic situation.
Education and empowerment of survivors
Involving patients in prevention, the follow-up and the management of late effects or the rehabilitation process (i.e. access to adapted information on self-management of late effects, on physical activities and dietary rules) is a major challenge. Satisfaction regarding the exchange of information with patients varied greatly among patients with low incomes (103). Online programs and e-health tools currently in development could help to improve the detection and evaluation of needs in supportive and palliative care (104,105). Online search for information, however, was lower for those who had lower education and socioeconomic status. Online programs and e-health may nevertheless offer a good alternative for educating survivors since they are considered costefficient and show equal impact with more conventional methods (106,107).
Education programmes are mainly targeted at specific subpopulations (e.g. patients with breast cancer, children, adolescents and young adults) but are also more effective for certain groups (e.g. white and well-educated patients, particularly women) (108).
Many programmes are implemented but not systematically evaluated. Costs and cost-effectiveness
are hardly addressed in evaluation studies (109).
Self-management
Cancer survivors are requesting a more active role in their health care. Self-management programmes need to be offered to cancer survivors and provide advices on how to look after themselves after cancer diagnosis, for example with adequate information about potential late effects and their early identification and management (110–115). Health-promoting measures, including web-based programs and telephone counselling, are attractive options to help patient to self-manage.
Perspectives in survivorship and rehabilitation cancer research
The literature shows that more data are needed concerning the different components of survivorship care. The main unanswered issues are:
- the impact of clinical follow-up on medical outcomes in a wide-range of cancers;
- the clinical, biological and cellular mechanisms of late-effects;
- the impact of supportive care and psychological support on quality of life (short and long term), survival, return-to-work;
- the long-term impact of education programmes;
- the long-term follow-up of adults surviving a childhood cancer,4 even 10, 20 and 30 years after end of treatment;
- the determinants of cancer inequalities linked to survivorship; and
- the impact and management of comorbidities and other health care disparities.
Furthermore, there is need for research networks and collaboration to initiate innovative clinical trials, such as intervention trials or RCTs.
Conclusions and recommendations
Based on the results obtained, the following section provides policy recommendations regarding three main aspects of the long-term follow-up care plans of cancer survivors: the content, and management and the implementation (Fig. 7.2).
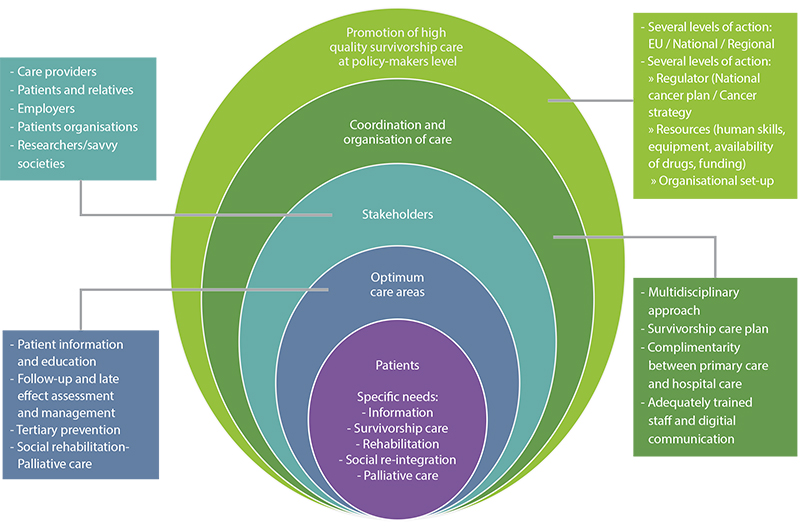
Fig. 7.2 Integrated survivorship and rehabilitation care model showing the different components of an effective European organizational survivorship care model with an integrated and personalized approach.
Medical follow-up and tertiary prevention
• An early and personalized follow-up programme should be systematically planned and delivered to each survivor.
• Baseline screening should be performed prior to the start of any cancer-specific treatment. After the first screening regular updates should be performed on an individual basis and followed by adequate provision of psychosocial care.
• An adequate and multidimensional assessment should be made of the survivor’s individual risk of late effects and respective rehabilitation or supportive needs (e.g. physical, psychological, social, cognitive, sexual, nutrition).
• Adequate and updated information on medium- and long-term effects of treatments should be available to survivors, their relatives and to care providers involved in the follow-up.
• In tertiary prevention, self-management should be emphasized, particularly on lifestyle recommendations and on avoiding risks of long-term effects by smoking cessation; weight control and healthy diet, including limited alcohol consumption; sufficient sustained physical activity at every phase of survivorship care; avoidance of excessive exposure to ultraviolet radiation; and stress management.
Patient-centred approach in long-term survivorship care
- Periodic screening of the physical condition, psychological distress and psychosocial needs should be conducted during the entire cancer pathway and integrated in routine cancer care.
- Rehabilitation and supportive care should be specifically offered to childhood, adolescent and young adult cancer survivors through a routine yearly psychosocial assessment with attention to social, psychological, behavioural and educational and/or vocational issues.
- Social and return-to-work issues should be integrated early into the cancer care pathway. The adaptation of working conditions for any patient returning to his/her previous work should be assessed at early stages.
- Public policies should be developed and implemented to safeguard cancer survivors’ working lives, their employability, skills and capacity to work, as well as their motivation to work. Selfemployed workers should be offered new skills to help them to achieve balance between health needs and work.
Management of long-term cancer follow-up care plans
- Psychosocial care, rehabilitation and palliative care should be integrated into the entire cancer pathway, including the survivorship and rehabilitation period. Psychosocial, rehabilitation and palliative care specialists should be members of (or associated with) the medical team in hospitals and in community care.
- The role of GPs and other PCPs should be actively supported to help them to manage all the care plan challenges. Their role should be clearly defined and tailored to the patient and the care plan needs. This role could evolve during the follow-up period.
- Communication between PCPs and health care specialists needs to be improved. Electronic patient records systems should be accessible to all health care providers treating patients. Communication between patients and health care providers needs to be improved.
- A key health care professional assuming a case management role should be assigned to each patient in accordance with medical and/or psychosocial specific requirements. This health care professional could play a main role in reducing the vulnerability of patients, for example with the management of adverse drug effects.
- Transition of care from paediatric oncology to adult medicine should be organized to guarantee adequate long-term follow-up and setting up of appropriate interventions.
- Empowerment of patients and their relatives should be enhanced to increase their participation in self-management, rehabilitation and return-to-work programmes. Online programs would facilitate this process together with the support of patient organizations.
- The use of digital methods (e-health supports) could facilitate sharing of information between patients and care providers and the uptake of the recommendations.
Perspectives in survivorship and rehabilitation cancer research
- An information and data collection system focused on late adverse effects (physical, psychological, cognitive, social and sexual), coupled with the surveillance of patients and involving PCPs, should be set up. More patient-reported outcome measures and their routine use are needed.
- The use of cancer registries in collecting data on survivors should produce stronger epidemiological data, including lifestyle, quality-of-life or socioeconomic information to better identify the causes of inequalities in survivorship. Moreover, registries should be expanded to additional factors that influence the quality of life (e.g. rehabilitation and employment issues). Patient-reported outcomes could also be a way to collect appropriate information.
- Clinical research should evaluate the feasibility, the efficacy and the cost-effectiveness (including the economical dimension) of non-drug related interventions such as self-management and e-health programs.
- Future research is needed to establish a multidimensional rehabilitation model focused on the quality of life and coordination of complex care to better address the management of late effects across the whole survivorship trajectory. More research would also be required to maximize the long-term follow-up and care of childhood cancer survivors and to identify the genetic risks associated with late effects and second cancers.
- More solid methodological RCTs and cohort studies are needed in order to reduce the intensity of cancer treatments while maintaining their efficacy, thus reducing the probability of late effects, particularly in childhood cancer survivors.
Implementation
Improving survivorship and rehabilitation care
- To enhance healthy lifestyles/behaviours in order to improve self-management of late effects with an equity perspective, public policies such as tobacco consumption restrictions, improving access to healthy food and developing actions to facilitate physical activity are needed. Actions targeted to specific vulnerable groups should be developed.
- Professional training and continuous education of health care providers is highly recommended for better information/communication/knowledge of survivorship and rehabilitation needs and management of late effects.
- Adequate financial and human resources should be allocated to the assessment and management of multidimensional late effects and tertiary prevention.
- Systematic screening of distress and physical and psychosocial needs is required for establishing adequate planning and implementation of psychosocial and rehabilitation care. A step-wise or tiered model of psychological care is recommended depending on the level of distress, psychological condition and morbidity of each patient. Interventions can range from basic information (level 1 and 2) to specialized psychological care (levels 3 and 4), as in the United Kingdom NICE guidelines. Interventions can range from:
- information and psycho-education by primary oncology team to peer support;
- e-health platforms for psychosocial support and self-management programmes;
- psychological interventions by psycho-oncology trained professionals (e.g. psychologists, social workers, psychiatrists);
- complementary spiritual support;
- psychotropic treatments by trained physicians (e.g. psychiatrists, oncologists); and
- patient support groups
For the diagnosis of psychological conditions, a specific assessment should be carried out by a psychological care professional: using validated and simple tools and according to clinical practice guidelines for the assessment and management of psychological distress and morbidity and anticipating the specific needs of populations at high risk, including young populations (e.g. children, adolescents, young adults) and relatives.
Education and self-management programmes should be developed and evaluated. Assessment of patients’ needs should be systematically part of the development of an education programme. The evaluation of these programmes should assess the impact on the personal, organizational and health care policy levels, including cost-effectiveness and impact on health care quality.
Professional experts networking on specific late effects (e.g. post-radiation neurotoxicity or drug-related impaired immune function) could facilitate their identification and management and support the GP. Those expert networks could also contribute to improve data collection and research on late effects. GP should contribute to the collection of these data.
Improving the management of survivorship and rehabilitation care
- Information and communications technology support, such as telemedicine or interoperative patient files, should facilitate patient management and follow-up.
- An information system should be implemented in order to monitor the activity of multidisciplinary teams including multiprofessional involvement, thus enabling the collection of information on process indicators such as coverage (e.g. the number of patients discussed compared with the number of patients under treatment) and implementation of survivorship care plans (e.g. survivorship care plan recommendations effectively implemented).
- Psychosocial and rehabilitation services need to be identified and provided for adequate referral of patients.
- Structural financial resources dedicated to psychological care, rehabilitation and social reintegration services should be embedded in the budgets of national or regional cancer care services. Inequalities of access to these services for underserved populations (e.g. people living in geographical and social isolation) have to be addressed.
- A financing mechanism prioritizing multidisciplinary over monodisciplinary interventions, already existing in some European health systems, can be used to integrate survivorship into the cancer care continuum. Psychological care and rehabilitation resources need to be available and in the private sector needs to be considered eligible for reimbursement from health insurance plans/companies.
- Economic evaluation should be undertaken in order to assess cost-effectiveness of the model of care and inform policy-makers for the most efficient use of resources.
Ensure the visibility and recognition of survivorship and rehabilitation on the policy agenda
- Health care system and patients benefit from the inclusion of survivorship and rehabilitation issues in national cancer control plans and policies. Partnership development across different professional groups, patients and cancer survivors is beneficial. Enhancing collaboration between the different representatives of these groups to support and optimize work with cancer survivors is advisable.
- It is important to involve patients in advocacy activities for the development of survivorship care, whether they are engaged in a formal organization or not.
- The implementation of a long-term follow-up policy for childhood, adolescent and young adult cancer survivors would improve their QALY, which will have a positive economic impact of reduction of direct (medical care, treatments) and indirect (sick leaves, incapacity of work) costs linked to long-term morbidities in this population. More international and multicentre cooperation could enhance research activities in this area because of the limited number of cases.
Ensure equal opportunities to all cancer patients
- Opportunities should be created for socially disadvantaged people to fully engage in follow-up programmes.
- Better access to self-management programmes should be available for underserved and deprived populations (low income/low education).
- Public policies should be developed and implemented to support cancer patients from diagnosis to return to work. This would include financial aspects such as access to loans, mortgages and life insurance. Implementation of a pan-European strategy to tackle the differences between workers with cancer in different countries and to prevent discrimination would generate more evidence to better understand the living conditions of cancer survivors who return to work.
- Employers can also play an important role in supporting the survivors’ return-to-work process: to explore possibilities of changes in job functions for cancer survivors and encourage them to acquire new skills; to facilitate the implementation of flexible working hours and options (remote working, part-time work); to offer economic benefits to employers who agree to adapt the workplace to the needs of cancer survivors; and to help self-employed workers to adapt their workplace and business to address health needs.
- Patient bills of rights including the right to work with special conditions (e.g. reduced hours of work or adapted working conditions) should be negotiated.
References
1 International Agency for Research on Cancer. GLOBECAN 2012: estimated cancer incidence, mortality and prevalence worldwide in 2012. Lyon, IARC Press; 2012 (http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx, accessed 14 January 2017).
2 EUROCARE. Conclusions of the MAC Roundtable “Key Determinants of Inequalities in Cancer Survival across Europe” (EP, 05 12. 2013). Rome, Istituto Superiore di Sanità; 2013 [National Institute of Health]; http://www.eurocare.it/Eurocare5/tabid/64/Default.aspx, accessed 14 January 2017).
3 Council of the European Union. 2876th Employment, Social Policy, Health and Consumer Affairs, Council meeting: conclusions on reducing the burden of cancer. Luxembourg, Employment, Social Policy, Health and Consumer Affairs Council; 2008 (http://www.eu2008.si/en/News_and_
Documents/Council_Conclusions/June/0609_EPSCO-cancer.pdf, accessed 15 January 2017).
4 Eurostat. Quality of life. Facts and views. Luxembourg: Publications Office of the European Union; 2015 (http://ec.europa.eu/eurostat/documents/3217494/6856423/KS-05-14-073-EN-N/742aee45-
4085-4dac-9e2e-9ed7e9501f23, accessed 14 January 2017).
5 Hewitt M, Greenfield S, Stovall E, eds. From cancer patient to cancer survivor: lost in transition. Washington, DC, US National Academy of Sciences; 2006.
6 Wolff SN. The burden of cancer survivorship. a pandemic of treatment success. In Feuerstein M, ed. Handbook of cancer survivorship. New York, Springer; 2006:7–18.
7 Earle CC. Quality of care. In Feuerstein M, ed. Handbook of cancer survivorhip. New York, Springer; 2006:19–42.
8 American Society of Clinical Oncology. ASCO cancer treatment and survivorship care plans. Alexandria, VA, American Society of Clinical Oncology; 2016 (http://www.cancer.net/survivorship/
follow-care-after-cancer-treatment/asco-cancer-treatment-and-survivorship-care-plans, accessed
14 January 2017).
9 Keesing S, McNamara, Rosenwax L. Cancer survivors’ experience of using survivorship care plans: a systematic review of qualitative studies. Journal of Cancer Survivorship, 2015;9:260–268.
10 Mullan F. Seasons of survival: reflections of a physician with cancer. New England Journal of Medicine, 1985;313(4):270–273.
11 US National Coalition for Cancer Survivorship. [web site]. Silver Spring, MD, National Coalition for Cancer Survivorship; 2017 (https://www.canceradvocacy.org/, accessed 14 January 2017).
12 Welch-McCaffrey D et al. Surviving adult cancers. Part 2: Psychosocial implications. Annals of Internal Medicine, 1989;111(6):517–524.
13 WHO. Rehabilitation. Geneva, World Health Organization; 2017 (http://www.who.int/topics/
rehabilitation/en, accessed 14 January 2017).
14 National Cancer Survivorship Initiative. Vision document. London, National Cancer Survivorship Initiative; 2008 (http://www.ncsi.org.uk/wp-content/uploads/NCSI-Vision-Document.pdf, accessed 15 January 2017).
15 National Cancer Institute. Follow-up care after cancer treatment. Silver Spring, MD, US National Cancer Institute; 2010 (https://www.cancer.gov/about-cancer/coping/survivorship/follow-upcare/
follow-up-fact-sheet, accessed 14 January 2017).
16 Lenihan DJ, Cardinale DM. Late cardiac effects of cancer treatment. Journal of Clinical Oncology, 2012;30(30):3657–3664.
17 Schmitz KH et al. Prospective surveillance and management of cardiac toxicity and health in breast cancer survivors. Cancer, 2012;118(Suppl):2270–2276.
18 Steingart RM et al. Cancer survivorship: cardiotoxic therapy in the adult cancer patient; cardiac outcomes with recommendations for patient management. Seminars in Oncology, 2013;40(6):690–708.
19 Kort JD et al. Fertility issues in cancer survivorship. CA: Cancer Journal for Clinicians, 2014;64(2):118–134.
20 Gosain R, Miller K. Symptoms and symptom management in long-term cancer survivors. Cancer Journal, 2013;19(5):405–409.
21 Pisu M et al. Dealing with the financial burden of cancer: perspectives of older breast cancer survivors. Supportive Care in Cancer, 2014;22(11):3045–3052.
22 Robinson KE et al. A quantitative meta-analysis of neurocognitive sequelae in survivors of pediatric brain tumors. Pediatric Blood Cancer, 2010;55(3):525–531.
23 Jacobs LA, Pucci DA. Adult survivors of childhood cancer: the medical and psychosocial late effects of cancer treatment and the impact on sexual and reproductive health. Journal of Sexual Medicine, 2013;10(Suppl 1):120–126.
24 Michel G, Auquier P, Long-term follow-up after therapy for childhood malignant hematologic disorder: the LEA program. Correspondances en Onco-Hématologie, 2011;6(1):42–48.
25 Magalhães JP. How ageing processes influence cancer. National Reviews of Cancer, 2013;13(5):357–365.
26 International Agency for Research on Cancer, WHO. Cancer prevention. In dos Santos Silva I, ed. Cancerepidemiology: principles and methods. Lyon, IARC Press; 1999:355–380.
27 Courneya KS, Karvinen KH, Vallance JKH. Exercise motivation and behavior Change. In Feuerstein M, ed. Handbook of cancer survivorship. New York, Springer; 2006:113–132.
28 Davies NJ, Batehup L, Thomas R. The role of diet and physical activity in breast, colorectal, and prostate cancer survivorship: a review of the literature. British Journal of Cancer, 2011;105(Suppl 1):S52–S73.
29 Ibrahim EM, Al-Homaidh A. Physical activity and survival after breast cancer diagnosis: meta‑analysis of published studies. Medical Oncology, 2011;28(3):753–765.
30 Hébert JR, Hurley TG, Harmon BE. A diet, physical activity, and stress reduction intervention in men with rising prostate-specific antigen after treatment for prostate cancer. Cancer Epidemiology, 2012;36(2):e128–e136.
31 Stevinson C et al. Exercise interventions for cancer patients: systematic review of controlled trials. Cancer Causes and Control, 2004;15(10):1035–1056.
32 Gil KM, von Gruenigen VE. Physical activity and gynecologic cancer survivorship. Recent Results Cancer Research, 2011;186:305–315.
33 Buffart LM et al. Evidence-based physical activity guidelines for cancer survivors: current guidelines, knowledge gaps and future research directions. Cancer Treatment Reviews, 2014;40(2):327–340.
34 Smith SH, Wu S. Persistent pain after breast cancer treatment. Annals of Palliative Medicine, 2012;1(3):182–194.
35 Ferrer RA et al. Exercise interventions for cancer survivors: a meta-analysis of quality of life outcomes. Annals of Behavioural Medicine, 2011;41(1):32–47.
36 Fong DY et al. Physical activity for cancer survivors: meta-analysis of randomised controlled trials. BMJ, 2012;344:e70.
37 Keogh JW, MacLeod RD. Body composition, physical fitness, functional performance, quality of life, and fatigue benefits of exercise for prostate cancer patients: a systematic review. Journal of Pain and Symptom Management, 2012;43(1):96–110.
38 Stagl JM et al. A randomized controlled trial of cognitive-behavioral stress management in breast cancer: survival and recurrence at 11-year follow-up. Breast Cancer Research and Treatment, 2015;154(2):319–328.
39 Powell ND, Tarr AJ, Sheridan JF. Psychosocial stress and inflammation in cancer. Brain Behavior and Immunity, 2013;30(Suppl):S41–S47.
40 Low CA, Beran T, Stanton AL. Adaptation in the face of advanced cancer. In Feuerstein M, ed. Handbook of cancer survivorship. New York, Springer; 2006:211–228.
41 Bruera E, Hui D. Conceptual models for integrating palliative care at cancer centers. Journal of Palliative Medicine, 2012;15(11):1261–1269.
42 Davis MP, Bruera E, Morganstern D. Early integration of palliative and supportive care in the cancer continuum: challenges and opportunities. American Society of Clinical Oncology Educational Book,
2013:144–150.
43 Abrahm JL. Integrating palliative care into comprehensive cancer care. Journal of the National Comprehensive Cancer Network, 2012;10:1192–1198.
44 Abernethy AP, Currow DC. Time for better integration of oncology and palliative care. Journal of Oncology Practice, 2011;7:346–348.
45 Osse B et al. Problems to discuss with cancer patients in palliative care: a comprehensive approach. Patient Education and Counselling, 2002;47:195–204.
46 Voogt E et al. Positive and negative affect after diagnosis of advanced cancer. Psychooncology, 2005;14:262–273.
47 Morasso G et al. Psychological and symptom distress in terminal cancer patients with met and unmet needs. Journal of Pain and Symptom Management, 1999;17:402–409.
48 Gilchrist LS et al. A framework for assessment in oncology rehabilitation. Physical Therapy, 2009;89(3), 286–306.
49 Mitchell AJ et al. Meta-analysis of screening and case finding tools for depression in cancer: evidence based recommendations for clinical practice on behalf of the Depression in Cancer Care Consensus Group. Journal of Affective Disorders, 2012;140(2):149–160.
50 Andersen BL et al. Screening, assessment, and care of anxiety and depressive symptoms in adults with cancer: an American Society of Clinical Oncology guideline adaptation. Journal of Clinical Oncology, 2014;32(15):1605–1619.
51 Victorson D et al. Measuring quality of life in cancer survivors. In Feuerstein M, ed. Handbook of cancer survivorship. New York, Springer; 2006:79–110.
52 Carvalho AF et al. Major depressive disorder in breast cancer: a critical systematic review of pharmacological and psychotherapeutic clinical trials. Cancer Treatment Reviews, 2014;40(3):349–55.
53 Nezu AM, Nezu CM. Psychological distress, depression and anxiety. In Feuerstein M, ed. Handbook of cancer survivorship. New York, Springer; 2006:323–338.
54 Raingruber B. The effectiveness of psychosocial interventions with cancer patients: an integrative review of the literature (2006–2011). ISRN Nursing, 2011:1–27.
55 Sheinfeld GS et al. Meta-analysis of psychosocial interventions to reduce pain in patients with cancer. Journal of Clinical Oncology, 2012;30(5):539–547.
56 Andrews L. Systematic review summary: psychosocial interventions to improve quality of life and emotional well-being for recently diagnosed cancer patients. Singapore Nursing Journal, 2013;40(3):47–49.
57 Grassi L, Travado L. The role of psychosocial oncology in cancer care. In Coleman MP et al., eds.
Responding to the challenge of cancer in Europe. Ljubljana, Institute of Public Health of the Republic of Slovenia; 2008:209–230.
58 Howell D et al for the Canadian Association of Psychosocial Oncology, Canadian Partnership Against Cancer. A pan-Canadian practice guideline: screening, assessment and care of psychosocial distress (depression, anxiety) in adults with cancer. Toronto, Canadian Partnership Against Cancer and the Canadian Association of Psychosocial Oncology; 2010.
59 Dauchy S et al. What psychological care should there be after cancer? Guidelines from the French Psycho-Oncology Society. Psychooncology, 2013;7:4–17.
60 Mitchell AJ, Vahabzadeh A, Magruder K. Screening for distress and depression in cancer settings: 10 lessons from 40 years of primary-care research. Psychooncology, 2011;20(6):572–84.
61 Mitchell AJ. Screening for cancer-related distress: when is implementation successful and when is it unsuccessful? Acta Oncologica, 2013;52(2):216–224.
62 National Health and Medical Research Council. Clinical practice guidelines for the psychosocial care of adults with cancer. Canberra, National Health and Medical Research Council; 2003 (https://www.nhmrc.gov.au/guidelines-publications/cp90, accessed 15 January 2017).
63 Tamagawa R et al. Who benefits from psychosocial interventions in oncology? A systematic review of psychological moderators of treatment outcome. Journal of Behavioral Medicine, 2012;35:658–673.
64 Travado L et al. Psychosocial oncology care resources in Europe: a study under the European Partnership on Action Against Cancer (EPAAC). Psychooncology, 2015; doi: 10.1002/pon.4044 (Epub ahead of print).
65 Bultz BD et al. President’s plenary International Psycho-Oncology Society: moving toward cancer care for the whole patient. Psychooncology, 2015;(12):1587–1593.
66 Mols F et al. Socio-economic implications of cancer survivorship: results from the PROFILES registry, European Journal of Cancer, 2012;48(13):2037–2042.
67 Mehnert A et al. Predictors of fear of recurrence in patients one year after cancer rehabilitation: a prospective study. Acta Oncologica, 2013;52:1102–1109.
68 Institut National du Cancer. La vie deux ans après un diagnostic de cancer. De l’annonce à l’après cancer, collection Etudes et enquêtes [Two years after a cancer diagnosis. From diagnosis to post cancer]. Paris, Institut National du Cancer; 2014 (http://www.e-cancer.fr/Expertises-et-publications/
Catalogue-des-publications/La-vie-deux-ans-apres-un-diagnostic-de-cancer-De-l-annonce-a-lapres-
cancer, accessed 14 January 2017).
69 Amir Z et al. Return to work after cancer in the UK: attitudes and experiences of line managers. Journal of Occupational Rehabilitation, 2010;20(4), 435–442.
70 Grunfeld EA, Low E, Cooper AF. Cancer survivors’ and employers’ perceptions of working following cancer treatment. Occupational Medicine, 60(8):611–617.
71 Bains M et al. Helping cancer survivors return to work: what providers tell us about the challenges in assisting cancer patients with work questions. Journal of Occupational Rehabilitation, 2012;22:71–77.
72 Nieuwenhuijsen K et al. Enhanced provider communication and patient education regarding return to work in cancer survivors following curative treatment: a pilot study. Journal of Occupational Rehabilitation, 2006;16:647–657.
73 Fong CJ et al. Psychological interventions to facilitate employment outcomes for cancer survivors: a systematic review and meta-analysis. Research on Social Work Practice, 2015:1–15.
74 Bottcher HM et al. Work-related predictors of not returning to work after inpatient rehabilitation in cancer patients. Acta Oncologica, 2013;52:1067–1075.
75 Feuerstein M et al. Work in cancer survivors: a model for practice and research, Journal of Cancer Survivorship, 2010;4(4):415–437.
76 Marino P et al. Sex differences in the return-to-work process of cancer survivors 2 years after diagnosis: results from a large French population-based sample. Journal of Clinical Oncology, 2013;12:77–84.
77 Moskowitz MC et al. Function and friction at work: a multidimensional analysis. Journal of Cancer Survivorship, 2014;8:173–182.
78 Thijs KM et al. Rehabilitation using high-intensity physical training and long-term return-towork in cancer survivors. Journal of Occupational Rehabilitation, 2012;22(2):220–229.
79 Van Muijen P et al. Factors associated with work disability in employed cancer survivors at 24-month sick leave, BMC Cancer, 2014;14:236.
80 Hoving J et al. Return to work of breast cancer survivors: a systematic review of intervention studies. BMC Cancer, 2009;9:117.
81 Munir F et al. Returning employees back to work: developing a measure for supervisors to support return to work (SSRW). Journal of Occupational Rehabilitation, 2012;22(2):196–208.
82 Torp S et al. Worksite adjustments and work ability among employed cancer survivors. Support Care Cancer, 2012(20):2149–2156.
83 de Boer AGEM et al. Interventions to enhance return-to-work for cancer patients. Cochrane Database of Systematic Reviews, 2015;(9):CD007569.
84 Alleweldt F. Study on the use of age, disability, sex, religion or belief, racial or ethnic origin and sexual orientation in financial services, in particular in the insurance and banking sectors. Berlin, Civic Consulting for DG Employment, Social Affairs & Inclusion, European Commission; 2010.
85 Kent EE et al. You’re too young for this : adolescent and young adults’ perspectives on cancer survivorship. Journal of Psychosocial Oncology, 2012;30:260–279.
86 Casillas J et al. How confident are young adult cancer survivors in managing their survivorship care? A report from the LIVESTRONG Survivorship Center of Excellence Network. Journal of Cancer Survivorship, 2011;5(4):371–381.
87 Metzger L et al. Female reproductive health after childhood, adolescent, and young adult cancers: guidelines for the assessment and management of female reproductive complications. Journal of Clinical Oncology, 2013;31:1239–1247.
88 Seitz, DCM, Besier T, Goldbeck L. Psychosocial interventions for adolescent cancer patients: a systematic review of the literature. Psychooncology, 2009;18:683–690.
89 Langeveld NE et al. Quality of life in young adult survivors of childhood cancer. Supportive Care in Cancer, 2002;10:579–600.
90 Sadak KT, Connor C, DeLuca H. Innovative educational approaches to engage and empower the adolescent and young adult childhood cancer survivor. Paediatric Blood Cancer, 2013;60:1919–1921.
91 Bingen K et al. Evaluation of a survivorship educational program for adolescents and young adult survivors of childhood cancer. Journal of Cancer Education, 2010;25:530–537.
92 Children’s Oncology Group. Long-term follow-up guidelines for surviv